Visible light photocatalytic degradation of pesticides and antibiotics byH3PO4-activated biochar combined with g-C3N4: Effects, mechanism, degradation pathway, and toxicity assessment. Shi, HJ (Shi, Haojie) [1] ; Wang, W (Wang, Wei) [1] ; Mao, LA (Mao, Liangang) [1] ; Zhang, L (Zhang, Lan) [1] ; Zhu, LZ (Zhu, Lizhen) [1] ; Wu, C (Wu, Chi) [1] ; Liu, XA (Liu, Xingang) [1]
Source JOURNAL OF ENVIRONMENTAL MANAGEMENT
DOI: 10.1016/j.jenvman.2025.124929
IF 8.4
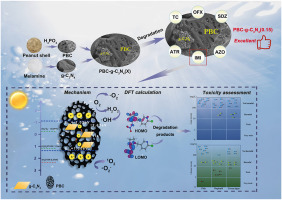
Abstract In recent decades, aquatic environmental pollution resulting from the extensive use of pesticides and antibiotics in the agriculture and livestock industries has become a major concern. In this study, a H3PO4 -activated biochar-g-C3N4 composite photocatalyst (PBC-g-C3N4 (0.15)) was synthesized for the degradation of pesticides (imidacloprid (IMI), atrazine (ATR), azoxystrobin (AZO)) and antibiotics (tetracycline (TC), ofloxacin (OFX), sulfadiazine (SDZ)) under visible light. PBC-g-C3N4 (0.15) achieved 89.7% degradation efficiency for IMI within 120 min, 1.8 times higher than g-C3N4, mainly due to 4.4 times increase in photocurrent density. Active species identified mainly included 1O2, ·O2−, and ∙OH. Density functional theory and UHPLC–Q Exactive MS analysis suggested three degradation pathways for IMI, with most intermediates predicted to be less toxic than the parent compound. Additionally, PBC-g-C3N4 (0.15) was largely unaffected by environmental factors and achieved over 94% degradation efficiencies for AZO, TC, and OFX within 60 min, and over 34% for ATR and SDZ in 120 min. The degradation efficiencies for IMI and TC remained 61.0% and 76.1% after eight cycles, respectively. In natural water, the degradation efficiencies for IMI and TC exceeded 30% and 65%, respectively. This research offers valuable insights into the modification of g-C3N4 and the treatment of pesticides and antibiotics in wastewater.
Graphical abstract